
In the Not Too Distant Future
April 8, 2024
We’re proud to present the full artwork for this year’s road trip! “Road to the Max” is going to be a wild ride you’ll not want to miss. This June we’re headed Down Under. Follow along as we tackle 1,800 km of Australia’s scenic east coast from Melbourne to Brisbane on our quest to discover the Sunburnt Country’s healthcare heroes.

The Max
April 5, 2024
A sneak peek at the artwork for this year's road trip. "Road to the Max" is scheduled to kick off on June 25!

Ford Falcon V8
March 18, 2024
We’ve secured our wheels for this year’s road trip — “Road to the Max.” Next month we’ll reveal the route we’ll take down the eastern Australian coast!

Road to Launch Episode 2
November 1, 2023
Episode 2 of 4 of The Road to Launch is here! With the crew fully assembled and our faithful vessel — Malilia — prepared, we were eager to set sail. If only we could have known then what the open ocean had in store for us. Head to PharmasAlmanac.com to see the second installment of our epic journey during the TransPac 2023 race!

Road to Launch Episode 1
November 1, 2023
Episode 1 of 4 of The Road to Launch is LIVE! We’ve got a boat, but now it’s time to get that boat secured for a 12-day voyage, assemble the crew, and begin training for whatever the Pacific Ocean might have in store for us. Head to PharmasAlmanac.com to see the first part of the captivating tale of our participation in TransPac 2023!

The Road to Launch
October 31, 2023
The cat is out of the bag. We’re finally unveiling the story of “Road to Launch”! The teaser dropped today, but stay tuned throughout the week for the four-part series as it rolls out on LinkedIn and on PharmasAlmanac.com.
It Keeps Going and Going...
September 18, 2023
With more than 10 years of social network building under his belt, we’re estimating that Nigel is less than three months away from breaking the 30,000 LinkedIn connections barrier. Pair that with Pharma’s Almanac’s 200,000+ strong database, and you can see why we’ve got one of the widest reaches in the industry.

Welcome, Danielle Alvarez!
January 28, 2022
The That’s Nice and Pharma’s Almanac teams are delighted to announce the introduction of Danielle Alvarez as the new Associate Scientific Editor for Pharma’s Almanac and Project Manager for That’s Nice. Danielle studied biology at Brown University and additionally brings marketing experience in support of the publication. Danielle is the first new addition in what will be a significant expansion of the Pharma’s Almanac team in 2022, further growing our resources in scientific content and market research under Editor in Chief David Alvaro. Danielle will support all aspects of our thought leadership platform, including interviews, articles, our Pharma’s Almanac TV video series, market research reports, and a number of new initiatives launching soon. Welcome aboard, Danielle!

David Alvaro, Chief Science Officer, Partner
January 25, 2022
David Alvaro has been a tremendous addition to the That’s Nice / Pharma’s Almanac team since joining in 2018. I’m personally proud to announce that David will be joining the Senior Management team as Chief Science Officer, Partner at That’s Nice, and Editor in Chief, Partner at Pharma’s Almanac. David’s Ph.D. was completed in 2008 in the Department of Genetics and Development at Columbia University Vagelos College of Physicians and Surgeons, and David is perfectly positioned to drive the scientific story for the Biotech and CDMO community that we serve at That’s Nice / Pharma’s Almanac. David’s team will be expanding in 2022 with an additional seven staff across all functional positions in scientific content and market research, setting the stage for the continued evolution of Pharma’s Almanac and That’s Nice. David will be joining Nigel Walker, Mark Allen, Wei Gao, Young Tae, and Yi Yi Liu as the sixth Partner at That’s Nice.

Welcome to That's Nice Danielle
January 3, 2022
Danielle Alvarez has joined the That's Nice team and will be working closely with the Pharma's Almanac publication. Starting 2022 off on the right foot.

Melanie and David Interview Tim
May 19, 2021
Tim Brewer from Yourway is back in front of the camera at the Yourway headquarters, Allentown PA.

Future of Healthcare 2020 Covers
December 18, 2020
2020 has been a great year to get a better understanding of the future of healthcare. Thank you Frank, Adam, Tim and Mark.

Q3 Pharma’s Alamanc Launches
September 25, 2020
We are very proud to have Adam Covitt on the cover of the Q3 Pharma’s Almanac. It's the last day in the London office before leaving for our 25-day tour.

An Interview with Tim Tyson
August 13, 2020
Nigel Walker interviews Tim Tyson on “The Future of Healthcare” and asks some difficult questions, fit for difficult times.

Pharma’s Almanac Interviews Globyz
August 12, 2020
Nigel Walker interviews Bryce Velletri, Project Director of Globyz, who provides insight on comparative souring.

Rhinebeck Scarlet Five Brand
July 28, 2020
The team is shooting the Q3 and Q4 creative ad for our Silver Jubilee campaign at the Old Rhinebeck Aerodrome. The motley band of fugitives made it happen!

A Fitting Cover, Continued Growth at TriRx
June 23, 2020
Tim Tyson and the management team continue to bring life back to the Huntsville, AL facility with tremendous growth over the past 9 months, and new hires starting every week. Congratulations to Tim and the team at TriRx. We are proud to have Tim Tyson on the cover of Q2 Pharma's Almanac.

Interview with David Alvaro
May 11, 2020
The interview with David Alvaro goes our for distribution on LinkedIn as we ramp up the interview list for Q3.

Staff Social Distancing
April 23, 2020
David Alvaro and Nigel met in Brooklyn today to discuss challenges with collecting enough information for the Q2 edition of Pharma’s Almanac. That's Nice is social distancing after 5 weeks of working at home.

Vibalogics, Oncolytic Viruses
April 20, 2020
The German-based Vibalogics is offering unique expertise and technologies to tackle next-generation manufacturing.

Kineticos Make It Happen
April 20, 2020
A very experienced team of consultants at Kineticos put out a solid article on next-generation medicines -- it's well worth the read.
Sosna + Co
April 20, 2020
The Sosnowski team gain traction in the past week with their licensing article from the Q1 edition of Pharma’s Almanac, which has enjoyed some great viewing numbers.

Tim Tyson, On What’s Possible
April 20, 2020
Just 4 weeks after the digital launch of the Pharma’s Almanac Q1 edition, Tim Tyson’s article rockets up to #4 in the rankings for most read on our LinkedIn network.

That’s Nice Supports Entrepreneurial Scotland
April 20, 2020
That’s Nice is happy to support the emerging biotech community with the guidance of the Entrepreneurial Scotland specialty management team.
OSE Immunotherapeutics
April 6, 2020
The Pharma's Almanac relationship with the biotech community has served the thought leadership team very well. The advanced medicines development by this community get more exciting each issue.
Growing Staff at Rentschler
April 6, 2020
I’m happy to see new talent arriving at Rentschler to support the growth of the CapX investment at Milford Mass. We look forward to continuing our support of the Rentschler team.

Supporting Biotechs
April 6, 2020
That’s Nice made a decision back in Q3 2019 to support (10) biotech each issue. We have now successfully covered 37 Biotech companies over the past 3 editions. I’m very pleased some of these companies have done well on the stock market.

Patrice Martin Leads the Way in Q1
March 30, 2020
Patrice Martin, General Manager of Servier CDMO drives through, leading with one of the most read articles in the Q1 Pharma's Almanac. Congratulations to the Servier team, there is some tough competition in this edition!

Albemarle Explains Small Volume Manufacturing
March 30, 2020
One of the original API manufacturing companies in the outsourcing space are adjusting to the changing demand in the market place.
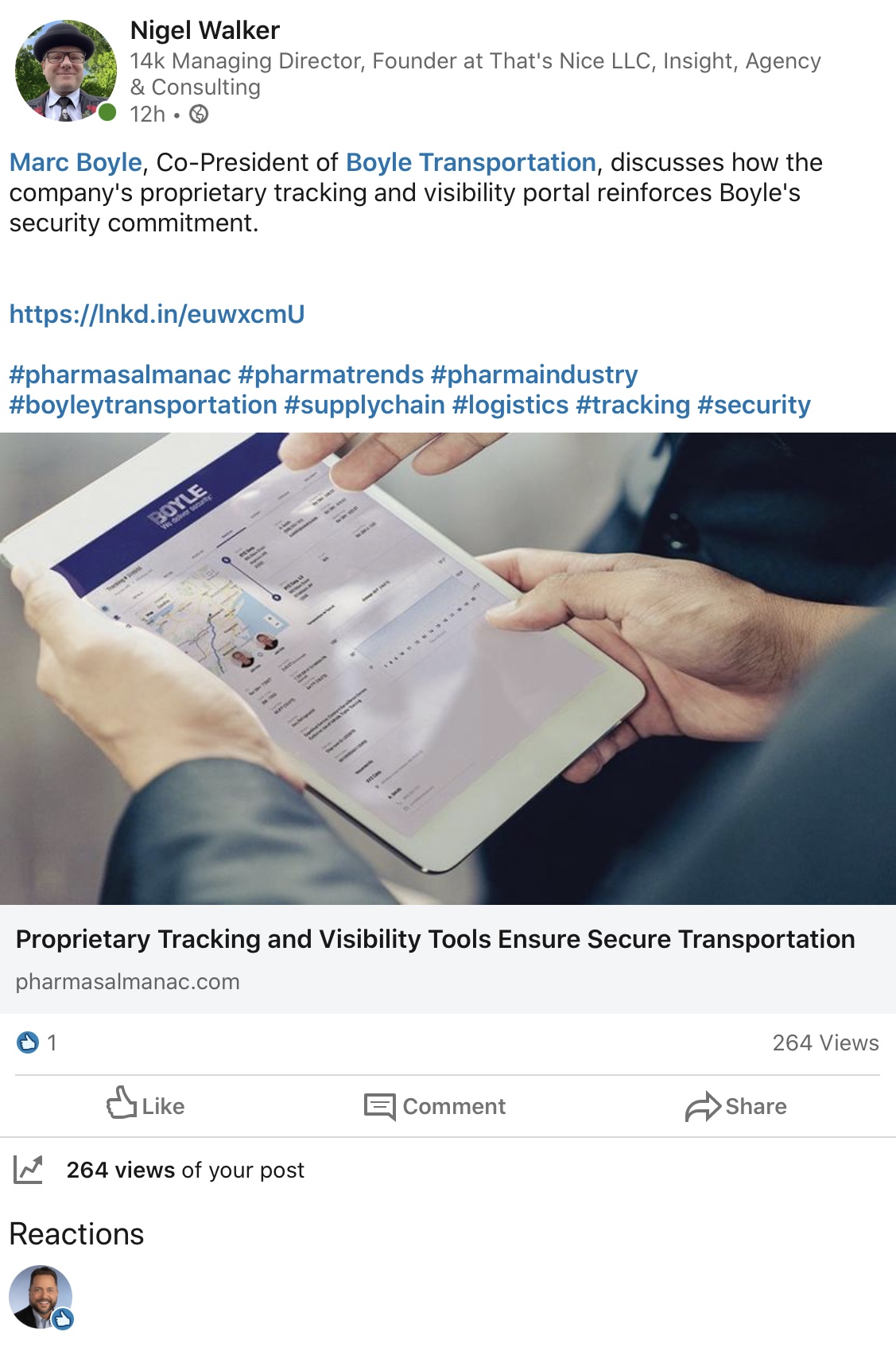
Valuable Covid-19 Transportation Materials
March 30, 2020
Boyle Transportation is continuing to make valuable deliveries for pharma and healthcare -- Andrew and Marc Boyle continue to make a difference in the industry.

Pii's Vision for a Dedicated CDMO
March 30, 2020
Kurt Nielsen, Ph.D. Discusses the future of Pii and how the vision for the CEO see’s the growth of a 26-year-old CDMO. That’s Nice are in full support of Pii as the values are supported and built upon.
Grifols Recombinant Protein Q1 Article
March 30, 2020
Grifols came out with another strong Biologics article in the Q1 Edition of PA. The team in Emeryville, CA are moving forward.
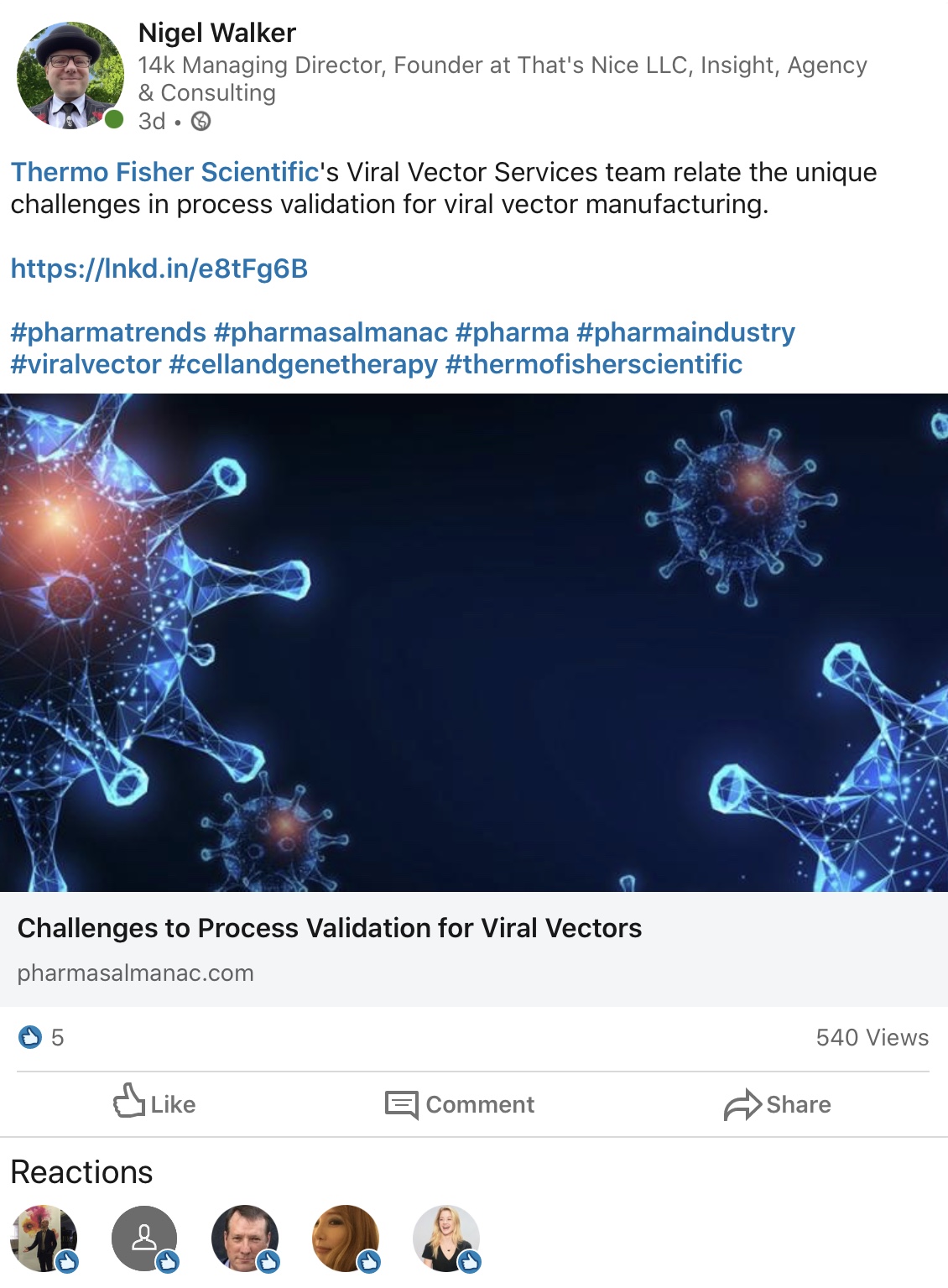
BrammerBio / ThermoFisher in PA Q1
March 29, 2020
ThermoFisher once again are pushing hard on the viral vector front, with expanded capacity coming online at their Lexington facility. We look forward to hearing more from Chris Murray.
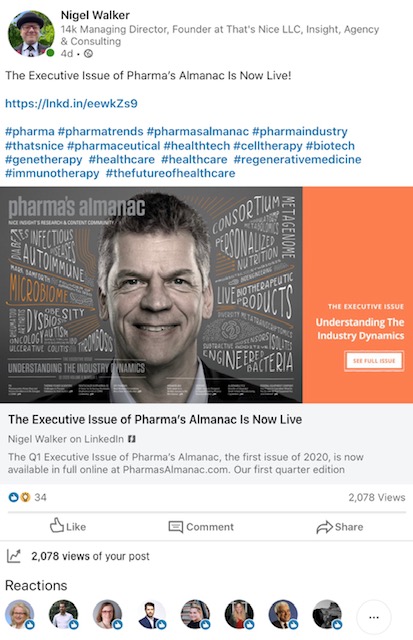
Q1 Pharma’s Almanac Mark Bamforth Cover
March 27, 2020
Mark Bamforth arrives on the cover of the Q1 edition of Pharma’s Almanac with a boom. The article has already had 2000+ views in the first 5 days from launch on LinkedIn, which is just one source . We are encouraged by the traffic from all 57 companies covered.

Our Favorite Cover of Pharma’s Almanac
March 19, 2020
Mark Bamforth, President and CEO of Arranta Bio, is featured on the cover of the Pharma’s Almanac Q1 Edition. It's our biggest issue ever and celebrates our Silver Jubilee.

Driving 2020
December 11, 2019
With lots going on at the end of year, Nigel's looking forward to plenty of driving in 2020.

Q4 Pharma's Almanac Issue Is Live
December 11, 2019
The Q4 Breakthrough edition of Pharma’s Almanac is live! The issue covers cell and gene therapy, as we look forward to another innovative year involving gene manipulation from the pharma industry.

PA Halftime
June 10, 2019
With the first two issues delivered and over 100 companies published to comprise 85% of our content, our B2B numbers speak for themselves.

BioVectra in Q2 Pharma’s Almanac - Booth #3055, 2427
June 3, 2019
Check out BioVectra’s must-see 8-page article in the Q2 Pharma’s Almanac.

CRB in Q2 Pharma’s Almanac - Booth #3205
June 3, 2019
CRB has led Pharma’s Almanac in most-read content for two years in a row.

Brammer Bio / ThermoFisher Booth #1501, 643, 1301, 2027
June 3, 2019
Brammer Bio is now part ThermoFisher! Come visit the Brammer Bio team at BIO International.

Fareva in Q2 Pharma’s Almanac - Booth #3455
June 3, 2019
Thibaut Fraisse gave a strong interview in the Q1 Pharma’s Almanac. Come by the Fareva booth to meet the team.

Aldevron Booth #4318
June 3, 2019
That’s Nice supports Aldevron, facilitating the company’s article on plasmids as part of a strategic content push in the Q2 issue of Pharma’s Almanac.

AbbVie Booth #526, 2927
June 3, 2019
If your haven’t yet, read the AbbVie article in Q2 Pharma’s Almanac, AbbVie is much more than just Humira.

The Advanced Therapeutics Issue Has Landed
May 24, 2019
Pharma’s Almanac, The Advanced Therapeutics Issue has landed on my desk - in my personal opinion, this issue has the best content.

Mark Bamforth in the Q2 Pharma's Almanac
May 13, 2019
We have a special article in the Q2 Pharma’s Almanac from one of our favorite Scots, Mark Bamforth. Mark will cover how he built two companies from the ground up over 7 years, subsequently selling them for $1.95 Billion - truly amazing!

Haig Barrett in the Q2 Pharma's Almanac
May 13, 2019
Our friends at Haig Barrett will be covered in the Q2 Edition of Pharma’s Almanac.

Polpharma Biologics in the Q2 Pharma's Almanac
May 13, 2019
Pharma’s Almanac will support Polpharma Biologics in the Q2 BIO International Edition.

Grifols in the Q2 Pharma's Almanac
May 13, 2019
There will be three business units of Grifols covered in the BIO International Q2 Edition of Pharma’s Almanac.

Catalent in the Q2 Pharma's Almanac
May 13, 2019
Pharma’s Almanac will support Catalent ODD business division for the first time in the Q2 BIO International Edition.